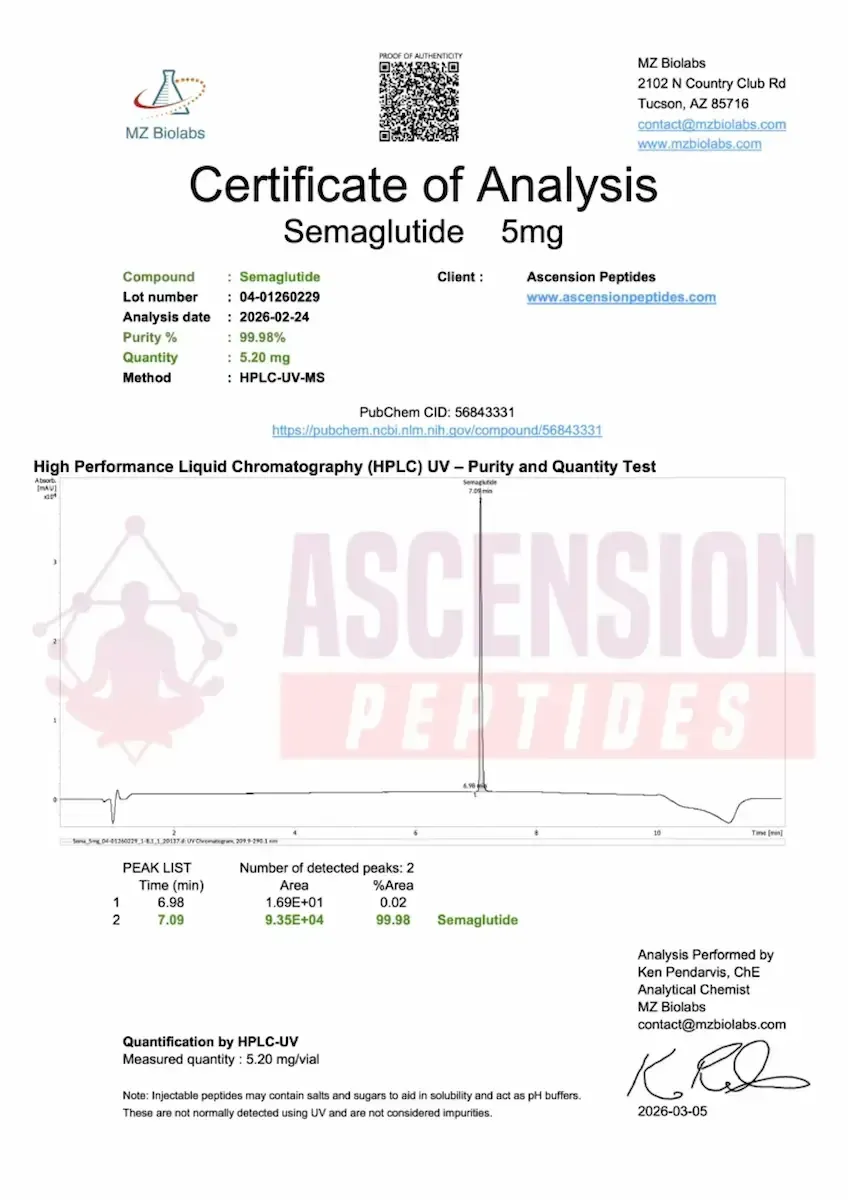
Independent U.S. third-party testing via HPLC-UV-MS Every batch is tested by MZ Biolabs (Tucson, AZ), an independent analytical laboratory with no operational connection to Ascension. Testing uses High-Performance Liquid Chromatography with UV detection and Mass Spectrometry (HPLC-UV-MS) to confirm both purity percentage and molecular identity. This dual-method approach catches impurities that UV alone may miss.
Public COA library with batch-specific documentation Certificates of Analysis are published on a dedicated page on Ascension's website. Each COA displays the lot number, analysis date, purity result, analyst name, and analyst signature. A recent Semaglutide batch (Lot 04-01260229, analyzed February 24, 2026) returned a verified purity of 99.98% via HPLC-UV-MS — performed by Ken Pendarvis, ChE, Analytical Chemist at MZ Biolabs. Results like this are representative of what Ascension posts publicly, not selectively.