The Best Peptide for Hormone Balance: Complete Guide to GH, Testosterone, and Endocrine Optimization
Peptide hormones are among the most precise signaling molecules in human physiology. They govern how the body releases growth hormone, regulates testosterone production, manages metabolic rate, and maintains the feedback loops that keep the endocrine system in equilibrium. Understanding how these molecules work is the foundation for evaluating which research peptides researchers and biohackers most commonly study in the context of hormone optimization.
This guide covers the biology of peptide hormones from first principles what they are, how they function, where they act and then moves into a structured comparison of the peptides most commonly associated with growth hormone release, endocrine support, and metabolic regulation.
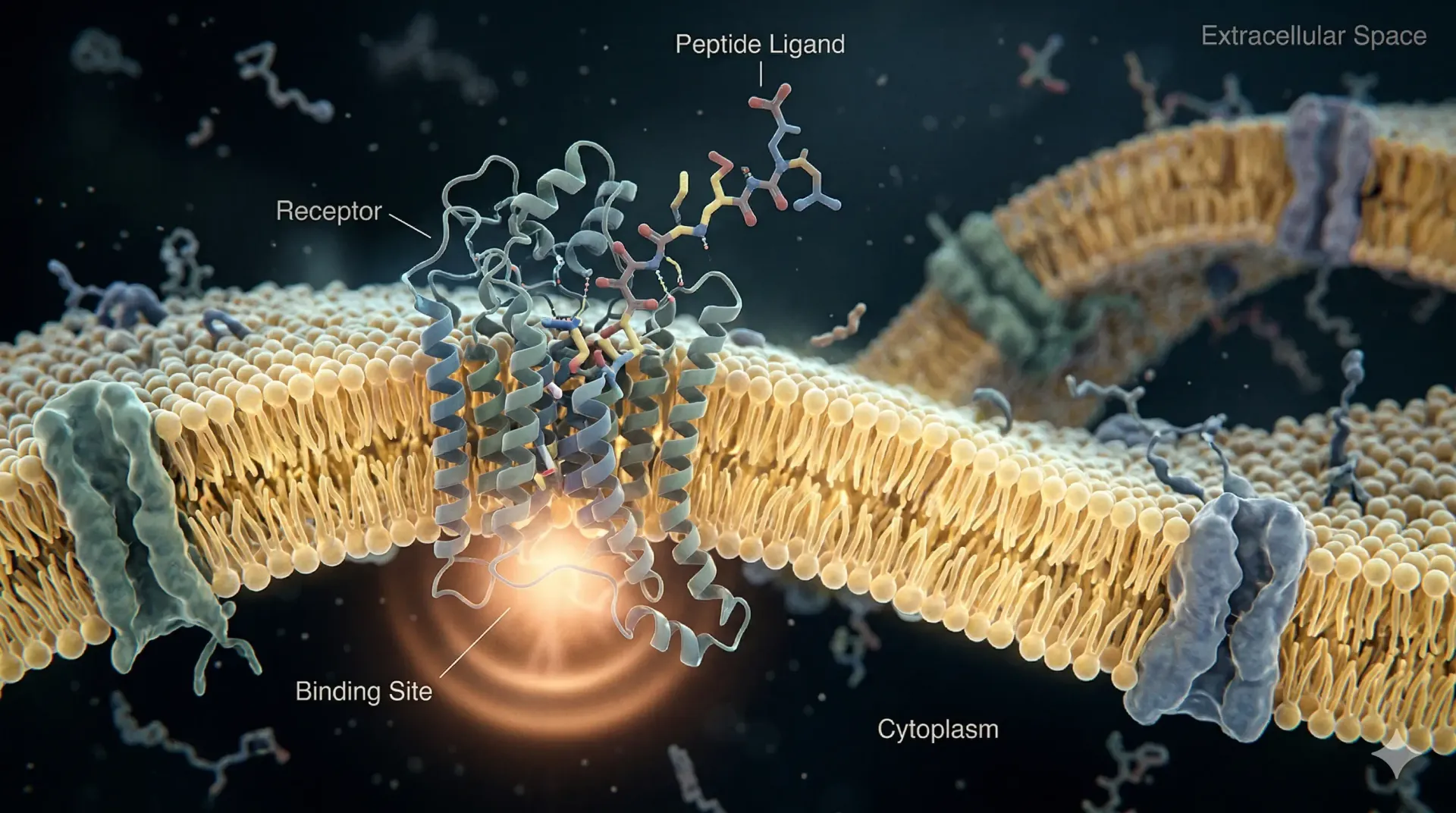
What Are Peptide Hormones
A peptide hormone is a signaling molecule made from a chain of amino acids. Unlike steroid hormones, which are derived from cholesterol and can pass through cell membranes, peptide hormones are hydrophilic. They do not cross the lipid bilayer of cells. Instead, they bind to receptors located on the cell surface and trigger a cascade of intracellular signals without ever entering the cell.
The body synthesizes peptide hormones in specialized cells within endocrine glands. The pituitary gland produces several of the most studied examples, including growth hormone (GH) itself. The hypothalamus produces releasing hormones, short peptides that signal the pituitary to secrete its own hormones. Insulin, glucagon, and ghrelin are also peptide hormones, each with distinct metabolic functions.
These molecules are short-lived in circulation. Enzymes in the blood rapidly degrade them, which is why peptide hormone signaling tends to operate in pulses rather than sustained surges. That pulse-based dynamic is directly relevant to how researchers think about exogenous research peptides and their interaction with the same pathways.
How Peptide Hormones Work in the Body
Peptide hormones work through receptor-mediated signal transduction. When a peptide hormone binds to its receptor on the surface of a target cell, it triggers a conformational change in that receptor. This change activates downstream signaling proteins most commonly G-proteins or receptor tyrosine kinases which in turn produce second messengers such as cyclic AMP (cAMP) or inositol triphosphate (IP3).
These second messengers carry the signal inside the cell, activating enzymes, altering gene expression, or triggering the release of stored substances. The effect of a single peptide hormone binding event can be amplified many times over through this cascade, which is why even small concentrations of peptide hormones produce measurable physiological changes.
Peptide hormone synthesis begins in the endoplasmic reticulum, where ribosomes translate mRNA into a prepropeptide a precursor molecule that includes signal sequences directing it through the secretory pathway. These sequences are cleaved as the peptide moves through the Golgi apparatus, where it is packaged into secretory vesicles and released into the bloodstream upon the appropriate stimulus.
Regulation of peptide hormone secretion occurs through negative feedback. When GH levels rise, for example, feedback signals to the hypothalamus and pituitary reduce the output of growth hormone-releasing hormone (GHRH) and increase the release of somatostatin, the inhibitory counterpart. This feedback architecture prevents runaway hormone production and keeps the endocrine system in dynamic balance.
Hormone Balance Explained
Hormone balance is not a fixed state. It is a dynamic equilibrium in which multiple hormones operate within ranges that support normal cellular function. The endocrine system is a network of feedback loops, and each axis, the hypothalamic-pituitary-adrenal (HPA) axis, the hypothalamic-pituitary-gonadal (HPG) axis, and the growth hormone axis interacts with the others.
Growth hormone influences insulin sensitivity. Testosterone levels affect cortisol responses. Cortisol suppresses GH secretion. Insulin modulates the activity of growth hormone receptors. These interactions mean that disruption in one axis often creates ripple effects across the others.
When researchers discuss peptides in the context of hormone balance, they are generally examining whether a given compound interacts meaningfully with one of these axes in a way that supports more stable signaling. The peptides most studied for this purpose target the GH axis, specifically the secretagogue pathway, which amplifies the pulsatile release of GH from the pituitary without introducing exogenous hormone directly.
Signs of Hormonal Imbalance
Hormonal imbalance presents across multiple systems. In the context of growth hormone and endocrine function, researchers have noted associations with the following symptom clusters in clinical populations: persistent fatigue unresponsive to sleep, increased adiposity particularly around the abdomen, reduced lean muscle mass, impaired recovery from physical stress, disrupted sleep architecture, reduced libido, mood changes including increased anxiety or flattened affect, and cognitive sluggishness sometimes described as brain fog.
These symptoms are nonspecific they overlap with many other conditions which is why endocrine evaluation typically involves laboratory testing of relevant hormone panels rather than symptom-based diagnosis alone. Understanding the underlying physiology is important context for anyone researching how peptide hormones relate to these presentations.
Best Peptides for Hormone Balance
Several research peptides have been extensively studied for their interactions with endocrine signaling pathways. The following are the peptides most commonly associated with hormone balance research, organized by mechanism and primary target.
Growth Hormone Secretagogues: Ipamorelin
Ipamorelin is a pentapeptide and selective growth hormone secretagogue. It mimics the action of ghrelin at the growth hormone secretagogue receptor (GHSR-1a), stimulating pulsatile GH release from the anterior pituitary. What distinguishes Ipamorelin from earlier secretagogues is its selectivity: it produces GH release with minimal concurrent elevation of cortisol, prolactin, or ACTH — hormones that can complicate the endocrine picture if elevated.
In research models, Ipamorelin has demonstrated dose-dependent increases in GH pulse amplitude without significantly affecting baseline GH secretion patterns between pulses. This selectivity makes it a frequently referenced comparator in studies examining the GH axis.
GHRH Analogues: CJC-1295
CJC-1295 is a synthetic analogue of growth hormone-releasing hormone (GHRH). While natural GHRH has a very short half-life due to rapid enzymatic degradation, CJC-1295 incorporates modifications that extend its activity significantly. The DAC (Drug Affinity Complex) version binds to albumin in the bloodstream, further extending its circulating half-life to several days.
CJC-1295 acts at the pituitary level rather than the hypothalamic level, directly stimulating somatotroph cells to release GH. When combined with a GHSR agonist like Ipamorelin, the two compounds act on complementary receptors and are frequently studied together as a synergistic pair. This combination targeting both the GHRH receptor and the ghrelin receptor simultaneously produces a more robust and sustained GH pulse than either compound alone in research settings.
Metabolic and Visceral Fat: Tesamorelin
Tesamorelin is a stabilized GHRH analogue that has undergone clinical investigation for its effects on visceral adiposity. It is the only compound in this class that has received regulatory approval for a specific indication reduction of excess abdominal fat in HIV-associated lipodystrophy which makes it one of the more thoroughly characterized research peptides in terms of clinical data.
Mechanistically, Tesamorelin stimulates the pituitary to release GH, which in turn activates lipolysis in adipose tissue. Elevated GH levels increase the activity of hormone-sensitive lipase, promoting the breakdown of stored triglycerides. The visceral adipose depot is particularly responsive to GH-mediated lipolysis, which is the basis for the research interest in Tesamorelin among those studying metabolic hormone balance.
Its use in research contexts is frequently discussed in relation to body composition, insulin sensitivity, and IGF-1 dynamics, all of which are downstream effects of GH axis modulation.
Systemic Support: BPC-157
BPC-157 (Body Protective Compound 157) is a synthetic pentadecapeptide derived from a protein found in gastric juice. It does not act directly on the GH axis, but it is frequently discussed in hormone balance research because of its proposed effects on systemic recovery, gut-brain signaling, and inflammatory regulation.
BPC-157 has been studied in animal models for its interactions with dopamine and serotonin receptor systems, nitric oxide signaling, and tendon and ligament repair pathways. Some researchers include it in hormone optimization discussions because inflammation and systemic stress have documented suppressive effects on GH secretion and testosterone production. Reducing that background load, in theory, allows the endocrine axes to operate with less interference.
It is categorized differently from the secretagogues above and is generally treated as a supportive rather than primary hormone-targeting compound in research protocols.
Mechanisms Behind Peptide Hormone Signaling
The distinction between receptor binding and intracellular signaling is central to understanding why peptide hormones behave the way they do. Because peptide hormones cannot cross the cell membrane, all of their biological activity depends on the receptor being present, functional, and appropriately coupled to its downstream effectors.
Receptor downregulation is a key concept here. Prolonged or excessive stimulation of a receptor can cause the cell to internalize and degrade those receptors, reducing its sensitivity to future stimulation. This is a documented feature of GH secretagogue pharmacology and is one reason why pulsatile rather than continuous stimulation is emphasized in the research literature.
Signal transduction through the cAMP pathway, the MAPK pathway, and JAK-STAT signaling (relevant to GH receptor activation) all converge on changes in gene expression. The effects observed hours after a GH pulse including increased IGF-1 production in the liver are the downstream result of these transcriptional changes.
Peptide Hormones vs Traditional Hormone Therapy
The distinction between peptide-based approaches and direct hormone replacement is meaningful and worth stating clearly. Exogenous hormone administration whether testosterone replacement therapy (TRT) or recombinant human growth hormone (rhGH) introduces the hormone directly into circulation, bypassing the body's own regulatory machinery.
Peptide secretagogues work upstream, stimulating the body's own glands to produce more of the target hormone. This preserves the feedback architecture of the endocrine system to a greater degree. The pituitary can still respond to negative feedback signals, which limits the degree to which any single peptide can override normal endocrine regulation.
This upstream mechanism also means that the effects of research peptides targeting the GH axis are inherently bounded by the functional capacity of the individual's pituitary. Someone with significantly impaired somatotroph function will not experience the same degree of response as someone with intact pituitary reserve.
Researchers interested in evaluating vendor quality for any of these compounds should review established quality verification criteria.
The Risks, Regulation, and Safety Considerations
Research peptides occupy a complex regulatory position. In the United States, compounds like Ipamorelin, CJC-1295, BPC-157, and Tesamorelin are not approved for general human use outside of specific clinical contexts (Tesamorelin being the exception under its approved indication). They are legal to purchase for research purposes but are not intended for human consumption under current FDA guidance.
From a safety standpoint, peptide hormones and their analogues interact with real physiological systems and carry real risk profiles. Elevated GH activity can affect insulin sensitivity, fluid retention, and potentially accelerate the growth of existing pathological cells. Any research into the endocrine effects of these compounds should be conducted with awareness of the relevant literature on both efficacy and adverse effects.
The absence of medical supervision in self-experimentation contexts introduces additional variables that complicate interpretation of outcomes. Researchers considering these compounds are encouraged to review published clinical literature, understand baseline endocrine status through laboratory testing, and be aware of the regulatory environment in their jurisdiction.
Third-party testing of research compounds is a minimum quality bar. Peptide purity, sterility, and accurate labeling are not guaranteed by default in the grey-market research space. The standards applied by reputable vendors are documented.
Who Benefits Most from Hormone-Regulating Peptides
Research into GH secretagogues and endocrine-targeting peptides has historically focused on several populations where the GH axis is most clearly affected by age or disease.
Adults over 40 experience a documented decline in GH pulse amplitude and frequency a process sometimes called somatopause. This decline tracks with changes in body composition, sleep architecture, and metabolic rate. Research in this population has examined whether GH secretagogues can partially restore more youthful GH pulsatility.
Men experiencing age-related testosterone decline present a different but related picture. Testosterone and GH interact through several mechanisms, including shared effects on lean body mass, fat distribution, and bone density. Some researchers study secretagogues in conjunction with other endocrine-targeted interventions to assess combined effects on anabolic signaling. For a deeper look at the relationship between testosterone and cellular energy systems, see our guide to testosterone and mitochondria.
Women navigating perimenopause and menopause face hormonal shifts that affect multiple axes simultaneously — estrogen, progesterone, GH, and cortisol all shift in ways that interact with each other. Research in this context is less developed than in men for most secretagogue compounds, though the underlying physiology of the GH axis does not differ fundamentally between sexes. Hair thinning is one of the most visible signs of these shifts, our guide to what peptide grows hair covers the peptides most studied for follicle restoration. Sexual dysfunction is another common hormonal consequence — our PT-141 dosage guide covers the melanocortin peptide most studied for desire and arousal.
Athletes and individuals focused on recovery and body composition represent a large segment of the research community interested in these compounds. The observed effects of enhanced GH pulsatility on collagen synthesis, skeletal muscle protein turnover, and fat metabolism make these peptides of interest for performance and recovery research.
RESEARCH RESOURCES
If you are researching peptides for hormone optimization, quality sourcing is the first variable to control. Review vendors in the Project Biohacking vendor directory and verify third-party testing practices before sourcing any research compound.
For reconstitution reference, the
peptide calculator provides a structured methodology for working with lyophilized peptides.
FAQ for Peptide Hormoness
What is the best peptide for hormone balance?
The peptides most studied for hormone balance are growth hormone secretagogues, particularly Ipamorelin and CJC-1295. Ipamorelin selectively stimulates pulsatile GH release with minimal side effects on cortisol or prolactin. CJC-1295 acts as a GHRH analogue, stimulating the pituitary to produce more GH. These compounds are research-use only and not approved for general human consumption.
Which peptide increases growth hormone the most?
CJC-1295 combined with Ipamorelin produces the most robust GH pulse in research models by acting on two complementary receptor pathways simultaneously. Tesamorelin is also a potent GHRH analogue with a well-characterized clinical dataset. Each compound has a distinct mechanism and duration of action.
Can peptides help with testosterone levels?
Peptide hormones do not directly stimulate testosterone production. Testosterone is primarily regulated through the HPG axis via luteinizing hormone (LH) and follicle-stimulating hormone (FSH). Some researchers study whether reducing systemic stress and improving GH pulsatility indirectly supports the hormonal environment in which testosterone production occurs, but a direct testosterone-elevating effect from secretagogues has not been established in research.
How do peptide hormones work in the body?
Peptide hormones bind to surface receptors on target cells and trigger signal transduction cascades inside the cell without entering it. These cascades produce second messengers like cAMP that activate downstream enzymes and alter gene expression. The result is a coordinated cellular response such as releasing stored GH that is amplified far beyond what the initial hormone concentration would suggest.
What is the difference between peptide therapy and hormone replacement?
Hormone replacement introduces the target hormone directly into circulation, bypassing the body's own regulatory mechanisms. Peptide secretagogues stimulate the body's own glands to produce more of the target hormone, preserving feedback architecture to a greater degree. This means secretagogues are bounded by the functional capacity of the underlying gland, while direct hormone replacement is not.
Do peptide hormones affect metabolism?
Yes. Growth hormone is a primary regulator of metabolism, influencing lipolysis, protein synthesis, glucose utilization, and IGF-1 production. Research peptides that stimulate GH release therefore have downstream metabolic effects. Tesamorelin has the most directly documented metabolic application, with clinical data showing reductions in visceral adipose tissue in studied populations.
Important Disclaimer: The content on Project Biohacking is for educational and informational purposes only and is not intended as medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional before making any changes to your health regimen, starting new supplements, peptides, or protocols. Nothing on this site establishes a doctor–patient relationship, and you use the information at your own risk. Research compounds discussed here are sold for laboratory research purposes only and are not approved for human or veterinary use or consumption.