About the Author:
Jeff Nunn is the founder of Project Biohacking. With over 30 years of biohacking practice, he applies decades of self-experimentation methodology to peptide research, dosing math, and vendor evaluation.
What the research actually shows about biliary and pancreatic risk with GLP-1 receptor agonists and how to interpret it.
GLP-1 receptor agonists were not designed with the gallbladder in mind. Their original mechanism of action targets the incretin system, slowing gastric emptying, suppressing glucagon, and increasing insulin sensitivity. Yet the biliary system is anatomically and functionally downstream of many of those same processes, and as clinical use of these drugs has scaled, reports of gallbladder-related complications have drawn increasing scrutiny.
The concern is not theoretical. A growing body of post-marketing data, alongside several large randomized controlled trials, has documented elevated rates of gallstones and related biliary events in patients using GLP-1 agonists. For a broader look at how GLP-1 and other peptides are used for weight management, see our guide to peptide therapy for weight loss. Understanding why requires looking at both the direct pharmacological effects and the indirect consequences of rapid weight loss, which these drugs reliably produce.
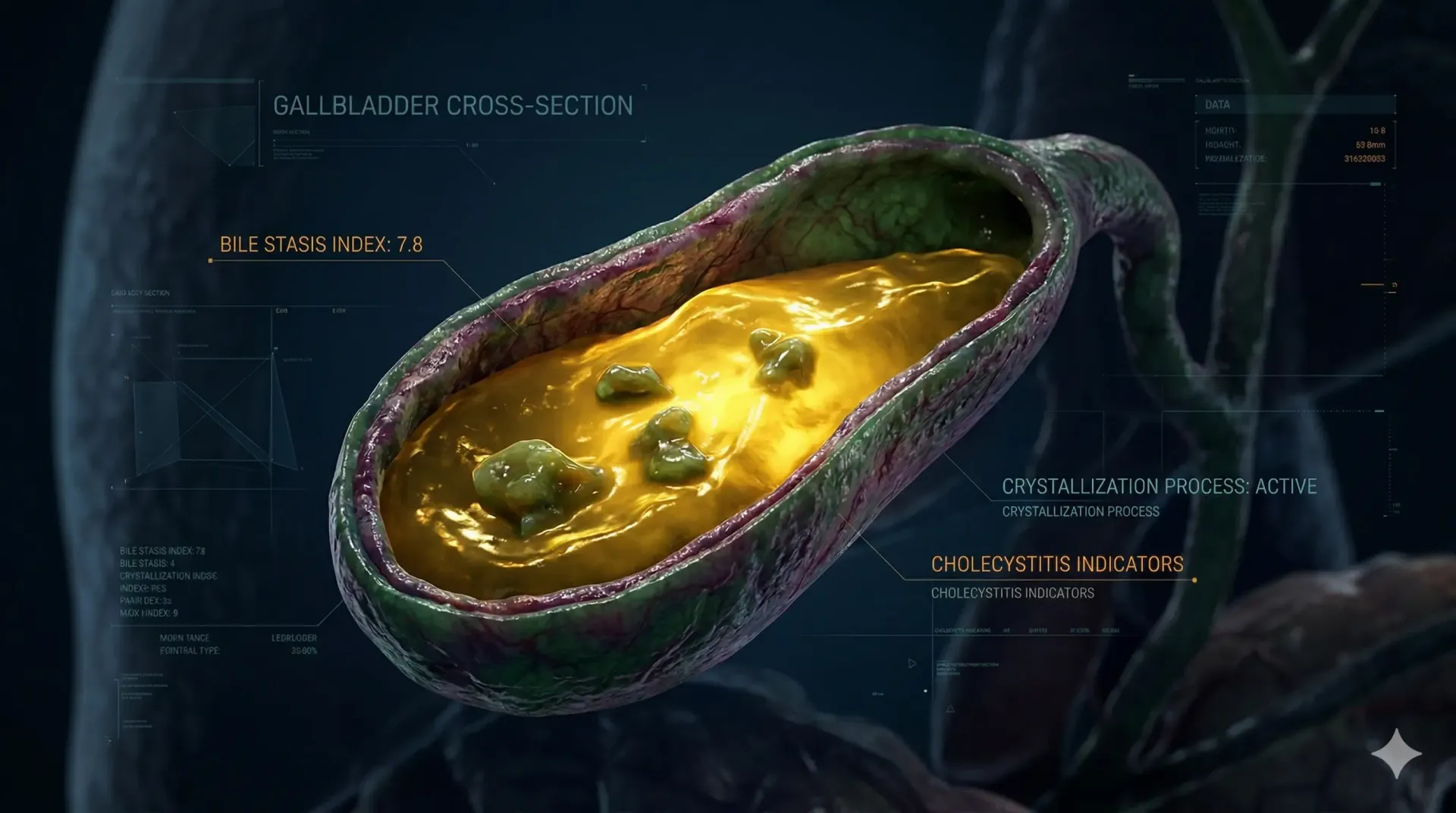
GLP-1 receptors are expressed not only in pancreatic beta cells and the gut, but also in the gallbladder wall. When GLP-1 receptor agonists activate these peripheral receptors, they appear to reduce gallbladder motility. In a normally functioning biliary system, the gallbladder contracts after meals to release bile into the small intestine, aiding fat digestion. GLP-1 agonist activity appears to blunt this postprandial contraction.
Reduced gallbladder motility means bile stays in the gallbladder longer between contractions. Bile that stagnates becomes more concentrated, and concentrated bile is more prone to forming sludge and eventually stones. This is the same underlying mechanism that explains why prolonged fasting, total parenteral nutrition, and very low-calorie diets also carry elevated gallstone risk. GLP-1 agonists appear to replicate some of that biliary stasis without requiring dietary restriction.
The weight loss component amplifies this dynamic. Rapid fat mobilization triggers an increase in biliary cholesterol secretion. When cholesterol output from the liver exceeds the capacity of bile acids and lecithin to keep it in solution, crystallization becomes more likely. The faster the weight loss, the higher the cholesterol load per unit of bile volume, and the greater the nucleation potential for stone formation.
For someone actively using a GLP-1 agonist, that mechanism is not an abstract physiology lesson—it is a concrete input into their personal risk curve. Before you escalate the dose or push weight loss harder, it is worth quantifying how those variables stack together in your case. That is exactly what the GLP-1 Risk Assessment Calculator is designed to do: integrate weight-loss velocity, dose intensity, and baseline biliary risk factors into a single, interpretable risk profile.
The SUSTAIN 6 and LEADER trials, both large cardiovascular outcomes studies, provided early signals that GLP-1 agonists were associated with higher rates of biliary events compared to placebo. These signals were modest but consistent across patient populations.
A more direct analysis came from the SCALE obesity trials with liraglutide, which reported a statistically significant increase in cholelithiasis in the treatment group. The hazard ratio was elevated even after adjusting for the degree of weight loss, suggesting that the drug itself, not just the weight reduction it induced, was contributing to gallbladder risk.
With semaglutide, the SUSTAIN and STEP trial series have shown similar patterns. A 2022 meta-analysis published in Alimentary Pharmacology and Therapeutics pooled data from multiple GLP-1 agonist trials and found a roughly 27 percent increase in cholelithiasis risk relative to control arms. The absolute risk remained relatively low, but the relative elevation was consistent across compound classes within the GLP-1 agonist family.
That kind of modest but consistent relative risk signal is precisely where structured decision tools add value. Instead of assuming “low risk” translates similarly for a 44-year-old woman with prior biliary sludge and rapid weight loss versus a leaner male on a cardiometabolic dose, you can explicitly weight those inputs. By walking through a series of targeted questions about age, sex, prior imaging, weight-loss trajectory, and dosing schedule, the GLP-1 Risk Assessment Calculator makes that nuance visible instead of leaving it buried in trial appendices.
Post-marketing surveillance data from the FDA Adverse Event Reporting System (FAERS) has captured increasing case reports of cholecystitis, cholelithiasis, and biliary pancreatitis in patients on GLP-1 agonists. These reports cannot establish causality on their own, but the signal volume has been sufficient to prompt label updates for several agents in this class.
A 2023 observational cohort study using insurance claims data compared patients initiating GLP-1 agonists against matched controls and found a statistically significant increase in acute cholecystitis, biliary colic, and cholecystectomy at 12-month follow-up. The effect was more pronounced in patients who experienced greater than 10 percent body weight reduction within the first six months of treatment.
Pancreatitis became a prominent safety topic for GLP-1 agonists shortly after the first agents in the class reached market. Preclinical data in rodents showed GLP-1 receptor activation could increase pancreatic duct cell proliferation. Case reports of acute pancreatitis in patients on exenatide and sitagliptin appeared in the early literature, and the FDA issued multiple safety communications requesting enhanced surveillance.
The concern was biologically plausible. GLP-1 receptors are expressed in the pancreatic ductal epithelium, and exocrine pancreatic function is closely tied to incretin signaling. The worry was that pharmacological overstimulation of this pathway could sensitize the pancreas to inflammatory triggers or directly promote acinar cell stress.
As the evidence base matured, the pancreatitis signal became more complicated to interpret. The LEADER trial, which enrolled over 9,000 patients with type 2 diabetes, found no statistically significant difference in acute pancreatitis rates between liraglutide and placebo. The SUSTAIN 6 trial with semaglutide showed a numerically higher rate of pancreatitis in the active arm, but the absolute numbers were small and the confidence intervals crossed unity.
A large meta-analysis published in the BMJ in 2020 examined pancreatitis rates across 189 randomized trials of GLP-1 agonists and DPP-4 inhibitors. The authors found no significant increase in pancreatitis risk attributable to either drug class compared to active comparators or placebo. The earlier signal, they concluded, was likely attributable to confounding: patients selected for GLP-1 therapy tend to have metabolic profiles, including obesity, hypertriglyceridemia, and alcohol use, that are themselves independent risk factors for pancreatitis.
The meta-analyses are reassuring at the population level, but they do not eliminate individual risk. Acute pancreatitis remains listed as a warning in the prescribing information for all approved GLP-1 agonists. Patients with a prior episode of pancreatitis, significant hypertriglyceridemia, or active gallstone disease represent a subgroup where the risk-benefit calculation looks different from the general population.
Biliary pancreatitis is worth distinguishing from idiopathic acute pancreatitis in this context. A gallstone that migrates into the common bile duct and obstructs the pancreatic duct can trigger pancreatitis by a purely mechanical mechanism. If GLP-1 agonists increase gallstone burden, they may indirectly elevate the risk of biliary pancreatitis even in patients with no direct pharmacological pancreatic sensitivity. This indirect pathway has received less systematic analysis than the direct GLP-1 receptor-mediated hypothesis.
Population-level risk elevation from GLP-1 agonists for biliary events appears real but modest. The absolute risk of developing clinically significant gallstones in any given patient depends heavily on their baseline risk profile.
Factors associated with higher gallstone risk in patients using GLP-1 agonists include: female sex, age above 40, prior history of gallbladder disease or biliary sludge on imaging, rapid early weight loss exceeding one percent of body weight per week, elevated baseline triglycerides, and use of high-dose or long-acting formulations. Patients with all of these factors represent a meaningfully different risk profile than a middle-aged male with modest weight loss goals and a clean prior abdominal imaging history.
If you recognize pieces of your own history in that higher-risk phenotype, the next step is not to abandon GLP-1 therapy altogether but to formalize your baseline risk estimate. A structured tool can help separate “this feels risky” from “this is measurably high risk once all variables are accounted for.” The GLP-1 Risk Assessment Calculator does this by scoring each of the major gallbladder and pancreatitis risk drivers and returning a stratified output that you can bring into a shared decision-making conversation with your clinician.
The rate of dose escalation may also matter. The incremental approach used in standard semaglutide and tirzepatide protocols reflects not only gastrointestinal tolerability concerns but also the recognition that slower metabolic adaptation may reduce the spike in biliary cholesterol secretion that accompanies rapid lipolysis.
Current evidence does not support routine prophylactic cholecystectomy or mandatory pre-treatment gallbladder imaging for all patients initiating GLP-1 therapy. However, baseline abdominal ultrasound is reasonable in high-risk individuals, particularly those with prior symptoms that were never fully evaluated or those with known metabolic risk factors for cholelithiasis.
Clinicians managing patients on GLP-1 agonists should be alert to right upper quadrant pain, particularly postprandial, which may indicate biliary colic before progression to acute cholecystitis or stone migration. Early recognition and intervention dramatically reduce morbidity from these outcomes.
On the pancreatitis side, patients should be counseled to report abdominal pain radiating to the back, particularly when it is persistent and associated with nausea or elevated amylase or lipase values on laboratory testing. The threshold for evaluation should be low in any patient on a GLP-1 agonist who presents with these symptoms, even if the population-level risk remains modest.
The public narrative around GLP-1 agonist safety has oscillated between dismissiveness and alarm, often without the nuance the data requires. The gallbladder signal is real, it is mechanistically coherent, and it has been replicated across multiple study designs. Acknowledging that does not make these drugs dangerous for most patients. It means their use should be thoughtful, individualized, and accompanied by appropriate monitoring.
The pancreatitis story illustrates the opposite failure mode. Early case reports created a durable public perception of elevated risk that subsequent large-scale data has substantially qualified. Patients and clinicians who rely on early signals without following the evolving evidence base will systematically miscalibrate their risk assessments in both directions.
The appropriate frame is not whether these drugs carry risk but whether that risk, contextualized against individual baseline factors, exceeds the well-documented benefits in metabolic disease, cardiovascular outcomes, and weight-related comorbidity reduction. For most patients in indicated populations, that calculus favors treatment. For the subset with high baseline biliary risk, it warrants additional monitoring and in some cases may influence drug selection or dosing strategy.
At this point, the mechanisms are clear, but the real question is how they apply to your specific situation.
Gallbladder risk with GLP-1 agonists is not binary. It scales based on how aggressively weight loss is occurring, how quickly doses are escalated, and whether baseline risk factors are already present.
Instead of trying to estimate that mentally, you can map it directly.
The
GLP-1 Risk Assessment Calculatorwas designed to model these exact variables so you can see where your risk profile actually sits.
Clinical trial data and post-marketing surveillance indicate that GLP-1 receptor agonists are associated with a modestly elevated risk of gallstone formation. The mechanism involves reduced gallbladder motility and, in patients experiencing rapid weight loss, increased biliary cholesterol secretion. The absolute risk depends heavily on individual baseline factors.
Rapid fat mobilization causes the liver to secrete more cholesterol into bile. When cholesterol output exceeds the capacity of bile acids and lecithin to hold it in solution, crystallization becomes more likely. GLP-1 agonists compound this by reducing postprandial gallbladder contraction, which concentrates bile between meals.
Early case reports raised concern about pancreatitis risk with GLP-1 agonists, but large randomized controlled trials and subsequent meta-analyses have not confirmed a statistically significant increase in risk at the population level. Patients with prior pancreatitis, high triglycerides, or active gallstone disease carry a different individual risk profile that warrants specific clinical consideration.
Biliary pancreatitis occurs when a gallstone migrates into the common bile duct and obstructs the pancreatic duct outlet. If GLP-1 agonists increase gallstone burden, they may indirectly elevate the risk of this form of pancreatitis even without direct pharmacological effects on the pancreas. This indirect pathway has received less systematic study than direct receptor-mediated hypotheses.
Patients with the highest risk profile include women over 40, individuals with prior gallbladder disease or biliary sludge, those experiencing rapid early weight loss, and patients with elevated baseline triglycerides. High-dose or long-acting formulations may also carry a higher risk profile than shorter-acting equivalents.
Current evidence does not support routine pre-treatment gallbladder ultrasound for all patients starting GLP-1 therapy. Baseline imaging is reasonable for individuals with prior biliary symptoms, known risk factors for cholelithiasis, or elevated metabolic risk profiles. Any patient developing right upper quadrant pain during treatment warrants prompt evaluation.

About the Author:
Jeff Nunn is the founder of Project Biohacking. With over 30 years of biohacking practice, he applies decades of self-experimentation methodology to peptide research, dosing math, and vendor evaluation.
Important Disclaimer: The content on Project Biohacking is for educational and informational purposes only and is not intended as medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional before making any changes to your health regimen, starting new supplements, peptides, or protocols. Nothing on this site establishes a doctor–patient relationship, and you use the information at your own risk. Research compounds discussed here are sold for laboratory research purposes only and are not approved for human or veterinary use or consumption.
“For educational use only. Not medical advice. Read our full disclaimer.”
+1 214-278-4039
All Rights Reserved | Project Biohacking