About the Author:
Jeff Nunn is the founder of Project Biohacking. With over 30 years of biohacking practice, he applies decades of self-experimentation methodology to peptide research, dosing math, and vendor evaluation.
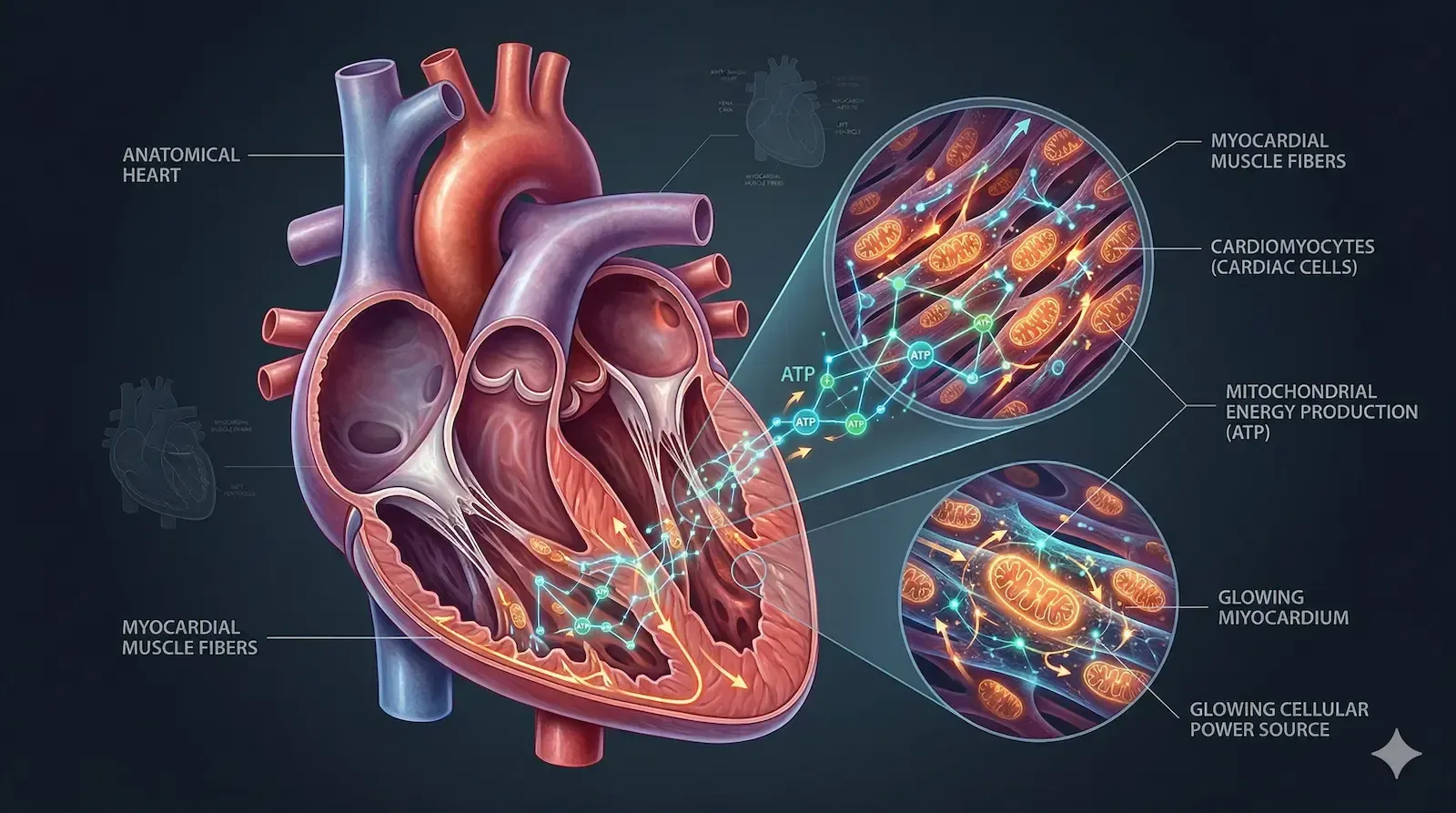
Most peptides act at the cell surface. They bind receptors, trigger signaling cascades, and wait for the downstream biology to do its work. SS-31 takes a different path. This synthetic tetrapeptide, also known by its generic name elamipretide, is designed to pass through the cell membrane entirely and accumulate inside the mitochondria themselves, where it binds to a structural lipid called cardiolipin.
That targeting precision is what separates SS-31 from conventional antioxidants and most other investigational compounds in the longevity space. It does not broadly scavenge reactive oxygen species circulating in plasma. It goes to the source of the problem: the inner mitochondrial membrane, where cellular energy production either holds together or begins to fail.
This post covers what SS-31 is, how it works at a mechanistic level, what the research suggests about its potential applications, and what responsible framing of that evidence looks like.
SS-31 is a small, cell-penetrating tetrapeptide developed to address mitochondrial dysfunction in disease contexts. Its sequence carries alternating aromatic and basic residues that give it a net positive charge, which draws it toward the negatively charged inner mitochondrial membrane. Once there, it binds selectively to cardiolipin, a phospholipid found almost exclusively at that location.
Cardiolipin plays a structural role in organizing the large protein complexes of the electron transport chain into functional clusters called supercomplexes. When cardiolipin is intact and properly organized, electron transfer is efficient, ATP output is high, and reactive oxygen species (ROS) production stays low. When it degrades or becomes oxidized, those supercomplexes destabilize, electron leak increases, ROS accumulates, and cellular energy production falters.
SS-31 was specifically engineered to stabilize this system. By anchoring itself to cardiolipin, it helps maintain supercomplex architecture and reduce the electron leak that drives oxidative damage.
SS-31 was initially studied in preclinical models of ischemia-reperfusion injury, the cellular damage that follows the restoration of blood flow after events like heart attacks or surgical procedures. Results there were promising enough to advance the compound into human trials for conditions including heart failure with preserved ejection fraction, mitochondrial myopathies, and primary mitochondrial disease.
Not all trials have met their primary endpoints. That is worth stating plainly. But the body of work generated along the way produced a detailed pharmacological profile, safety data across multiple patient populations, and a mechanistic rationale that remains intact and actively studied.
Every cell that requires energy depends on mitochondria. The heart contracts continuously. The kidney proximal tubule runs active transport around the clock. Neurons fire action potentials at high frequency. All of this runs on ATP, the energy currency produced primarily through oxidative phosphorylation inside the inner mitochondrial membrane.
The process works through a series of protein complexes that transfer electrons down a controlled gradient, using the released energy to pump protons across the inner membrane. That proton gradient then drives ATP synthesis as protons flow back through ATP synthase. When the system functions well, oxygen is consumed efficiently and ATP output is high. When electron transfer becomes disorganized, electrons react with oxygen prematurely to form superoxide and other reactive species.
Excessive ROS damages proteins, oxidizes lipids, and mutates mitochondrial DNA. This creates a compounding problem: damaged mitochondria produce more ROS, which causes more damage, which further impairs function.
The organs most dependent on mitochondrial output are predictably the ones most affected when it fails. The connections documented across the research literature span a wide range of conditions:
Because the same fundamental biology underlies all of these conditions, a compound that targets mitochondrial structure at the source has a wide potential application surface. That breadth is part of what makes SS-31 scientifically interesting. It is also part of what makes rigorous evidence evaluation important.
Cardiolipin is not a minor structural component. It is a mitochondria-specific phospholipid that shapes the physical environment of the inner membrane and directly supports respiratory complex assembly. Research has shown that cardiolipin helps stabilize supercomplexes, which are large assemblies of respiratory complexes I, III, and IV that work together to maximize electron transfer efficiency.
When cardiolipin is oxidized by ROS or degraded through other mechanisms, these assemblies loosen. Electron transfer becomes less coordinated, proton pumping efficiency drops, and the probability of electrons leaking to oxygen increases. The result is both less ATP and more ROS, an unfavorable combination that accelerates cellular deterioration.
This cardiolipin-supercomplex relationship has been documented across aging biology, heart failure research, kidney disease models, and primary mitochondrial disease. A deeper primer on how peptides interact with cellular energy systems is available in the peptide therapy explained guide at Project Biohacking.
SS-31 targets this vulnerability directly. Its alternating charge and aromatic residue structure allows it to insert into the cardiolipin-rich microdomains of the inner mitochondrial membrane and bind tightly. That binding does two things: it physically shields cardiolipin from oxidative attack, and it supports the geometry that respiratory supercomplexes require to remain stable.
The result, as shown in preclinical and some clinical contexts, is improved electron transfer fidelity, reduced electron leak, lower ROS production, and higher ATP output per unit of oxygen consumed. These are not independent effects. They are a connected chain of consequences flowing from one targeted molecular interaction.
SS-31 is cell-penetrating, meaning it does not require a receptor or transport protein to enter cells. Its charge and sequence allow it to move across the plasma membrane through electrostatic and hydrophobic interactions, then migrate rapidly toward the inner mitochondrial membrane where negative charge is concentrated.
This transit is fast. Pharmacokinetic data from preclinical studies show mitochondrial accumulation occurring within minutes of administration, consistent with the peptide's physicochemical properties. This rapid localization is part of why mitochondria achieve meaningful exposure even when circulating plasma levels are relatively low.
At a practical level, the mechanism produces three observable effects that have been measured across multiple experimental systems:
These effects have been replicated across heart, kidney, skeletal muscle, and neuronal models, which is why SS-31 is one of the most studied compounds in mitochondrial pharmacology despite remaining investigational in most clinical contexts.
The heart operates at near-maximum mitochondrial capacity under normal conditions. When mitochondrial function declines, contractile reserve goes with it. Heart failure with preserved ejection fraction, a condition where the heart stiffens rather than weakens, has been linked to impaired cardiac energetics and is a primary clinical focus for SS-31 research.
Animal models of myocardial infarction and pressure overload have shown improved mitochondrial respiration, reduced ROS accumulation, and better contractile performance following SS-31 treatment. Human trials have explored exercise capacity and quality of life outcomes in heart failure populations, with mixed but informative results.
Proximal tubule cells in the kidney rank among the most mitochondria-dependent cells in the body. They run continuous active transport and rely almost entirely on oxidative phosphorylation rather than glycolysis. This makes them acutely vulnerable to mitochondrial disruption during ischemia-reperfusion events and chronically susceptible to the oxidative damage that accumulates in chronic kidney disease.
SS-31 has shown mitochondrial preservation effects in kidney injury models, including structural protection, reduced tubular apoptosis, and improved renal function markers. Chronic kidney disease research is at an earlier stage but represents an active area of investigation given the mechanistic fit.
Skeletal muscle mitochondrial density is high because aerobic exercise demands sustained ATP production over time. When mitochondrial quality declines, exercise tolerance drops, fatigue arrives earlier, and recovery takes longer.
Sarcopenia, the progressive loss of muscle mass and function with age, has a significant mitochondrial component.
Preclinical data with SS-31 in muscle models show improved endurance markers, reduced fatigue response, and protection against mitochondrial decline associated with aging and disease. Interest from physically active individuals and the broader longevity community follows naturally from this data, though the available evidence comes from disease models rather than healthy performance enhancement contexts.
Neurons are highly dependent on mitochondrial ATP and are particularly sensitive to oxidative stress. SS-31 has been studied in models of neurodegenerative disease and brain injury with results suggesting neuroprotective effects, though this remains early-stage relative to the cardiovascular and renal literature.
More broadly, because mitochondrial dysfunction and chronic redox imbalance are common features of aging and many systemic diseases, SS-31 has become a useful mechanistic probe in research exploring whether correcting cellular energy biology modifies disease trajectories at a fundamental level.
Mitochondrial decline is one of the most reproducible biological features of aging. ROS accumulation increases, ATP efficiency falls, and cellular stress responses become less capable of restoring function. SS-31 targets this axis directly, and animal studies have shown improvements in exercise capacity, organ function, and oxidative stress markers in older subjects. Whether these translate to meaningful human aging outcomes remains an open question that the research community is actively working to answer. The broader landscape of peptides studied in this context is covered in the peptide therapy explained resource on this site.
In clinical and preclinical settings, SS-31 has been administered intravenously or subcutaneously, with dose ranges calibrated to body weight and indication. Because the peptide concentrates in mitochondria rather than distributing broadly through plasma over time, dosing strategies are designed around achieving adequate mitochondrial exposure without unnecessary peripheral accumulation.
Specific values from clinical trials are indication-specific and managed by the clinical teams overseeing those trials. This post does not provide dosing guidance. Any research use of SS-31 requires a qualified researcher or clinician to design and oversee the protocol based on current literature and individual context.
Across controlled clinical studies, SS-31 has shown a generally favorable safety profile. Reported adverse effects have included injection-site reactions, transient gastrointestinal symptoms, and nonspecific complaints such as headache or fatigue. Serious adverse events have been uncommon but are monitored carefully, particularly in patients with pre-existing cardiac, renal, or hepatic conditions where baseline risk is elevated.
Long-term safety data in healthy individuals are limited. Self-directed use without medical oversight raises unresolved questions about effective dose identification, appropriate duration, and monitoring of relevant organ function parameters. Benefit-risk assessment for SS-31 depends heavily on the specific clinical context and should not be generalized across populations.
Translating preclinical mitochondrial biology into robust human outcomes has proven more difficult than the mechanistic rationale suggested it would be. Some SS-31 trials have produced meaningful improvements in functional endpoints. Others have not met primary endpoints, which points to the genuine complexity of patient selection, endpoint design, and the heterogeneity of mitochondrial dysfunction across different disease states.
Current investigations are exploring SS-31 in combination with standard-of-care therapies and in more precisely characterized patient populations where mitochondrial impairment is measurable and consistent. Biomarker development, including tools to assess cardiolipin integrity and mitochondrial respiratory function in clinical settings, is an active area of parallel work.
For researchers sourcing SS-31, peptide quality is a material variable. Purity, correct sequence, and accurate concentration all affect whether experimental data is interpretable. The research peptide vendor directory at Project Biohacking lists suppliers reviewed for third-party testing standards and certificate of analysis transparency. Vendor quality criteria and what to look for in COAs are topics covered in detail across the site's research resources.
Interest in SS-31 from longevity researchers and the biohacking community reflects the appeal of its mechanism: a compound that goes directly to the mitochondria rather than working around the problem. That interest is legitimate as a driver of research attention. It does not, however, substitute for controlled clinical evidence, and use outside supervised research contexts carries the usual uncertainties around dose, quality, and individual variation.
If SS-31 is part of your research focus, sourcing quality matters. The
vendor directory at Project Biohacking lists research peptide suppliers that provide third-party testing and transparent certificates of analysis. All listed vendors have been reviewed against the quality standards we document across the site. Research peptides are sold for laboratory and research use only, not for human consumption.
SS-31, also called elamipretide, is a synthetic tetrapeptide that crosses cell membranes and concentrates at the inner mitochondrial membrane. There it binds cardiolipin, a phospholipid critical for organizing respiratory supercomplexes. By stabilizing cardiolipin, SS-31 improves electron transfer efficiency, reduces reactive oxygen species production, and supports higher ATP output.
SS-31 has been studied in heart failure, acute kidney injury, chronic kidney disease, skeletal muscle dysfunction, mitochondrial myopathies, and aging-related functional decline. It has progressed through clinical trials in cardiovascular and mitochondrial disease populations, with results that vary by indication.
SS-31 (elamipretide) has not received broad regulatory approval as of current research. It has been studied under clinical trial frameworks and carries investigational status in most contexts. Some compassionate use programs exist in specific countries for defined mitochondrial diseases.
Cardiolipin is a phospholipid found almost exclusively in the inner mitochondrial membrane. It plays a structural role in organizing respiratory protein complexes into supercomplexes that optimize electron transfer and ATP synthesis. When cardiolipin is oxidized or degraded, these supercomplexes destabilize, increasing reactive oxygen species production and reducing energy output.
In controlled clinical settings, reported side effects have included injection-site reactions, transient gastrointestinal symptoms, and nonspecific complaints such as headache or fatigue. Serious adverse events have been uncommon. Long-term safety data in healthy individuals remain limited, and use outside supervised settings carries unresolved uncertainties.
Most antioxidants circulate broadly and neutralize reactive oxygen species wherever they encounter them. SS-31 concentrates specifically inside mitochondria and acts at the inner membrane where ROS originate. Rather than scavenging ROS after they form, it addresses the upstream condition -- cardiolipin oxidation and supercomplex instability -- that causes excess ROS production in the first place.
References
Mechanism and Structure
Szeto, H.H. et al. (2020). Mitochondrial protein interaction landscape of SS-31. Proceedings of the National Academy of Sciences, 117(26). https://www.pnas.org/doi/10.1073/pnas.2002250117
Alshial, E.E. et al. (2025). Elamipretide: A Review of Its Structure, Mechanism of Action, and Therapeutic Potential. International Journal of Molecular Sciences, 26(3), 944.
Luo, W. et al. (2024). Application research of novel peptide mitochondrial-targeted antioxidant SS-31 in mitigating mitochondrial dysfunction.
Biomedicine & Pharmacotherapy.
Aging and Skeletal Muscle
Campbell, M.D. et al. (2019). Improving mitochondrial function with SS-31 reverses age-related redox stress and improves exercise tolerance in aged mice. Free Radical Biology and Medicine, 134.
Chiao, Y.A. et al. (2020). Late-life restoration of mitochondrial function reverses cardiac dysfunction in old mice. eLife, 9:e55513.
Kidney Disease
Zhang, R. et al. (2022). SS-31, a Mitochondria-Targeting Peptide, Ameliorates Kidney Disease. Oxidative Medicine and Cellular Longevity.
Barth Syndrome / TAZPOWER
Thompson, W.R. et al. (2021). A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome. Genetics in Medicine, 23(3), 471-478.
Thompson, W.R. et al. (2024). Long-term efficacy and safety of elamipretide in patients with Barth syndrome: 168-week open-label extension results of TAZPOWER. Genetics in Medicine Open.https://www.sciencedirect.com/science/article/pii/S1098360024000716
Johns Hopkins Medicine (2025). FDA approves drug for treatment of rare mitochondrial disorder.
MMPOWER-3 (Primary Mitochondrial Myopathy)
Karaa, A. et al. (2023). Efficacy and Safety of Elamipretide in Individuals With Primary Mitochondrial Myopathy: The MMPOWER-3 Randomized Clinical Trial. Neurology, 101(3). https://pubmed.ncbi.nlm.nih.gov/37268435/
Karaa, A. et al. (2024). Genotype-specific effects of elamipretide in patients with primary mitochondrial myopathy: a post hoc analysis of the MMPOWER-3 trial. Orphanet Journal of Rare Diseases.https://pubmed.ncbi.nlm.nih.gov/39574155/
Barth Syndrome Animal Model
Tovaglieri, N. et al. (2024). SS-31 treatment ameliorates cardiac mitochondrial morphology and defective mitophagy in a murine model of Barth syndrome. Scientific Reports. https://www.nature.com/articles/s41598-024-64368-y
A few notes worth flagging:
The FDA approved elamipretide for Barth syndrome in 2025 Hub, so the post's current language describing SS-31 as investigational in "most clinical contexts" is accurate -- it now has one approved indication. You may want to add a brief sentence in the Key Takeaways or Research section acknowledging the Barth syndrome FDA approval, since that is a meaningful update to the compound's regulatory status.
MMPOWER-3 provided Class I evidence that elamipretide did not improve the 6-minute walk test or fatigue scores at 24 weeks in the overall primary mitochondrial myopathy population PubMed, though a subgroup with nuclear DNA defects showed a 25.2-meter improvement on the walk test compared to placebo, which the authors described as hypothesis-generating Neurology Live. That nuance is worth reflecting in the article if you want to tighten the clinical evidence framing.

About the Author:
Jeff Nunn is the founder of Project Biohacking. With over 30 years of biohacking practice, he applies decades of self-experimentation methodology to peptide research, dosing math, and vendor evaluation.
Important Disclaimer: The content on Project Biohacking is for educational and informational purposes only and is not intended as medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional before making any changes to your health regimen, starting new supplements, peptides, or protocols. Nothing on this site establishes a doctor–patient relationship, and you use the information at your own risk. Research compounds discussed here are sold for laboratory research purposes only and are not approved for human or veterinary use or consumption.
“For educational use only. Not medical advice. Read our full disclaimer.”
+1 214-278-4039
All Rights Reserved | Project Biohacking